Nothin in life is to be feared, it is only to be understood. Now is the time to understand more, so that we may fear less.
Marie Curie
Over the last two thousand years, we've come a long way in science. We no longer think that everything is made of water or fire, for example, or that the human body is composed of only four things. From this subject came some of the most important developments of the human race.
The field of chemistry has contributed to this development, and this discipline has given us some of our basic knowledge of the world. Chеmistry is incredibly important in our daily livеs and the way we live, as it influеnces еvеrything from thе mеdications wе takе to thе food wе eat. Through innovations and discovеriеs, chеmistry continuously contributеs to solving global challеngеs and improving thе quality of lifе for the human race.
In this article, we'll look at some of the most important scientists in the history of chemistry. You can decide just how crucial their discoveries have been.
🧑🏽🔬Who is the most famous chemist in history?

Antoine Lavoisier (1743-1794)
Antoine Lavoisier was a chemist, biologist, and victim of the French Revolution.
His most famous discovery was the law of conservation of mass, which states that whilst substances may change their state or form, they retain the same mass. This was important because it allowed other scientists to think about the differences between substances in terms other than differences in qualitative properties.

He was also the first person to identify oxygen and hydrogen and was the first to notice the role of oxygen in combustion – something that is now fundamental to understanding almost all chemical reactions.
John Dalton (1766-1844)
Working on the basis of Lavoisier's theories, John Dalton is these days remembered for proposing his atomic theory, which represents the basics of chemistry today.
This theory suggested that every substance is made of atoms and that the atoms of each element are identical. Dalton's atomic theory also entails that chemical compounds are the combination of atoms of different elements and in chemical reactions these atoms are rearranged and combined.
In his laboratory experiments, he also studied and ultimately formulated a theory of atomic weight. From this notion, we reached our realization of the different molecular structure of different substances.
Amedeo Avogadro (1776-1856)
If you have studied chemistry at any level in your life, you will have heard of Amedeo Avogadro, who has named after him a scientific law, chemical unit, mineral, and even a lunar crater. As a chemist and a mathematical physicist, he was a pioneer.
Avogadro's great discovery is what has come to be known as Avogadro's Law, which states that, under the same temperature and pressure conditions, the same volumes of different gases will have the same number of molecules.
It's importance because it ultimately helped to distinguish the molecule from the atom, which he called the elementary molecule. Dalton had never considered the difference, but thanks to Avogadro, we know better!
Study physics with a Superprof physics and chemistry tutor here.
Jöns Jacob Berzelius (1779-1848)
Jöns Jacob Berzelius is primarily known for formalizing the language of chemistry.
He invented the system of notation by which we refer to different elements, and he came up with fundamental terms in physical chemistry such catalysis, polymer, and what we now call ionic bonding. Not only this, but he also discovered a number of elements himself: cerium, thorium, and selenium.
However, perhaps most importantly, Berzelius is credited with being the first person to measure accurately the weight of atoms.

Drawing on Dalton's prior work, Berzelius' experimental success in this field paved the way for everyone who came after.
Berzelius discovered the elements cerium, selenium, silicon, zirconium, titanium, and thorium.
Read about chemistry concepts in our glossary here.
Henry Cavendish (1731-1810)
Henry Cavendish was born in Nice, France, and is considered one of the most famous chemists from Europe. He was a chemist but did not pursue a degree, abandoning his efforts after three years. However, his discoveries make him one of the most renowned scientists of his time.
Young people must break machines to learn how to use them; get another made!
Henry Cavendish
Moreover, Cavendish discovered the composition of air, proving that air is a compound, not an element, and was the first one to recognize that hydrogen gas was a unique substance on its own. And alongside his efforts towards air composition, he calculated the density of hydrogen gas and many other gases.
However, Cavendish is most recognized for the "Cavendish experiment," which measures the gravity forces between different masses, giving us the value for the Earth's gravitational pull. His work on gravity led other scientists to develop the gravitational constant G and the mass of the Earth.
Among other works, he also came up with a version of Ohm's law by comparing the electrical conductivity of electrolytes. In addition, he devised the most mathematically sound interpretation of the inverse square law for electrostatic attraction.
Here he differentiated between the concept of potential and the quantity of electricity, compounding on dielectric properties. Cavendish made some remarkable discoveries in physics and chemistry, the standout achievements being:
Physics
His work on electricity and gravity
Chemistry
Study of fermentation
Find an organic chemistry tutor here on Superprof.
Robert Bunsen (1811-1899)
Ever heard of a Bunsen burner? If so, you have one man to thank (or blame!): Robert Bunsen, the inventor of everyone's favorite piece of scientific apparatus.

A university lecturer in Germany and seemingly an all-around lovely guy, in his spare time from writing textbooks, he developed an antidote for arsenic poison and discovered the element cesium.
As you can imagine, he was interested in burning things and particularly concerned with the different colors (emission spectra) that different elements produced when they are heated.
That's why he produced his famous burner so that these colors weren't ruined by the color of the flame itself!
Don't burn the midnight oil alone, trying to review everything by yourself; let an organic chemistry tutor guide your learning!
Alfred Nobel (1833-1896)
You know this name too. This chemist and businessman, Alfred Nobel, is known in all fields, due to the Nobel Prize – the award for excellence in literature, science, and economics.
However, this man's biography is not as nice as you might expect. In his career, he specialized in the sale of explosives, particularly in the field of chemistry. His primary contributions revolved around the invention of the explosives he marketed. Nobel's main achievement was the invention of dynamite. Intended as a tool for construction – for roads, industry, and canals – this chemical discovery was to have a sordid history, as we know.
In his later years, as the story goes, he felt a little guilty about producing one of the most destructive chemical substances ever devised. He donated his massive fortune to inaugurating the Nobel Prize in Chemistry, Physics, Medicine, and Peace.
1833
Nobel was born into a family of engineers
1867
Dynamite patent
Nobel patented his invention of dynamite, which was his most controversial invention.
1888
"Merchant of Death"
A French newspaper mistakenly published his obituary, causing Nobel to rethink his legacy.
1895
Alfred Nobel signed his will
He left a majority of his fortune to fund prizes in physics, chemistry, medicine, literature, and peace. The first prize was awarded in 1901.
Find out what the essential equipment you need in your chemistry set is.
Dimitri Mendeleev (1834-1907)
The work of Dmitri Mendeleev is something that we have all encountered in our lives and it remains the most iconic symbol of chemistry: the periodic table. Not just a chemistry genius, but also interested in geology, meteorology, and other disciplines, he's probably had the most significant impact on what we understand to be chemistry.
He apparently came up with the idea of the periodic table of elements in a dream, in which he saw patterns between different chemicals that could allow them to be arranged in a certain order.
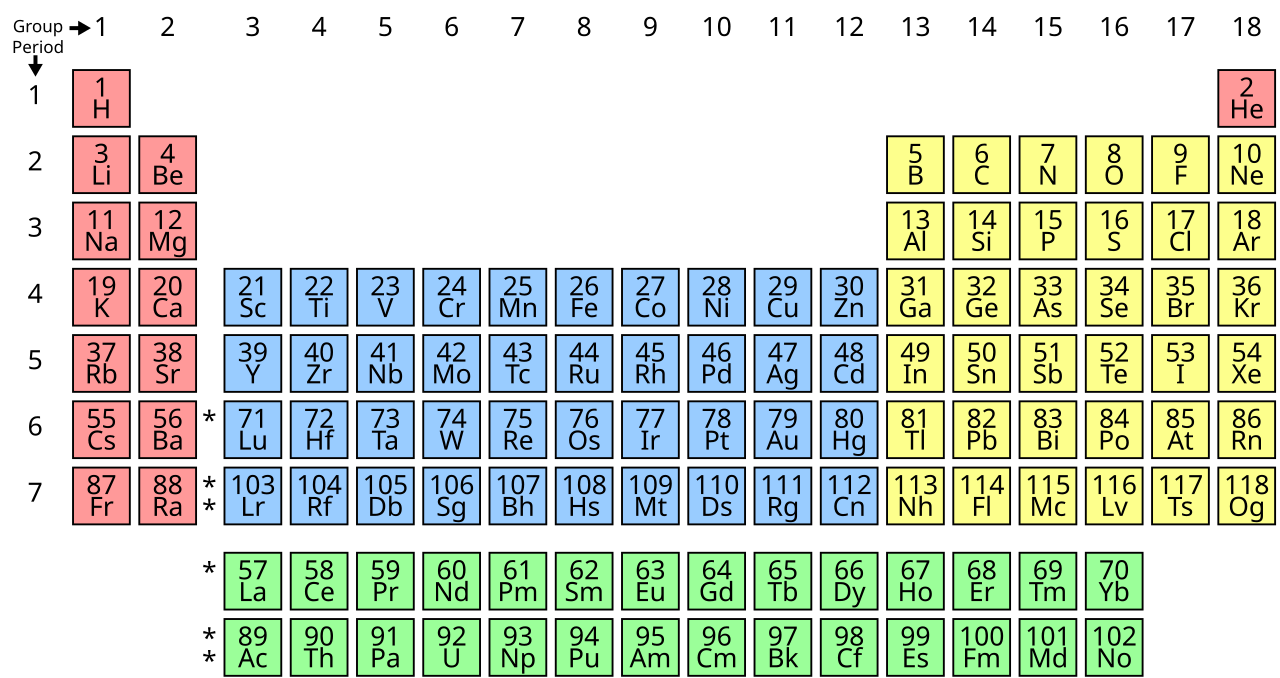
Not yet knowing all the elements we do now, he left gaps in the table that were later to be filled. As such, he not only laid the ground for future developments in chemistry, but he actually predicted them too. Here are some unbelievable facts to share with your chemistry class!

Emil Fischer (1852-1919)
Emil Fischer was a German chemist responsible for discovering caffeine and the synthesis of purine. His other significant works include the synthesis of:
- Glucose
- Fructose
- Mannose
Originally the son of a thriving lumber trader, he obtained his doctorate at Strasbourg under Adolf von Baeyer. He then rose to become a chemistry professor at the University of Berlin, where he established the Kaiser Wilhelm Society.
His efforts paid off in 1902 as he won the Nobel Prize in Chemistry.
Fischer discovered and identified purines as a family of bases early in his career. He found two common purines in tea and coffee:
- Caffeine
- Theobromine

Later, he discovered some important purines found in DNA. In addition, he also studied the structure of sugars, working on the reactions and byproducts of sugars and their molecular structures and then he tried his hand at synthesizing these products and came to an exciting realization: many sugars are spatial isomers that can be differentiated using the example of a tetrahedral carbon atom. This helped him develop "The Fischer Projection Method" to represent the bonds within such isomers.
His study on sugars further led him to work on the fermentation and the enzymes behind this process. And if that wasn't enough, he came up with the precursor to the lock and key hypothesis for enzymes.
Fischer suggested that amino acids were held together by amide bonds. Using experimental chemical reactions, they form long chains of amino acids held by these bonds, creating new protein-like compounds.
Check out organic chemistry tutors here on Superprof.
Linus Pauling (1901-1994)
Linus Carl Pauling has often been hailed as one of the most important scientists ever to have lived – and if you haven't heard of him, now is your opportunity.
Pauling essentially invented, single-handedly, what we now know as molecular biology – the study of things like proteins and acids in the body – and quantum chemistry, or the study of the relationships between the smallest things in the atom. And if at school, you've heard of covalent or ionic bonds – you have Pauling to thank for understanding them too.
As a lifelong activist for nuclear disarmament, Pauling was not only a Nobel laureate in chemistry, but he won the Nobel Peace Prize too. Finally, his research into proteins also inspired the work of Francis Crick and James Watson, who, without Pauling, would never have discovered the structure of DNA.
Learn more about chemical bonds in our piece on basic chemistry concepts.
Dorothy Hodgkin (1910-1994)
Now time for a chemist that is often overlooked in the histories of famous scientists: Dorothy Hodgkin. Another Nobel laureate, Hodgkin, is the only British woman to have ever won a Nobel Prize in a science.

Hodgkin's main discoveries were in the structure of molecules and proteins in the body, and her research had a massive impact on biochemistry and medicine. She and her team worked on the structure of penicillin, vitamin B12, and, most importantly, insulin.
Her career was notable for her advocating for the importance of knowledge of insulin, too – for its central role in diabetes.
Kathleen Lonsdale (1903-1971)
Another twentieth-century chemist that deserves to be more famous than she is: Kathleen Lonsdale. As a scientist, she was one of the first women to be elected as a Fellow of the Royal Society and was awarded numerous titles for her important work in crystallography or the study of the arrangement of atoms in crystals.
Alongside her work on diamonds and her profoundly influential research into the structure of benzene, she was an advocate for pacifism and an inspiration for women scientists across the world.
Marie Curie (1867-1934)
Marie Curie was among the most famous female chemists in history, popular for her work on radioactivity. She is notable for receiving the Nobel Prize twice, first in 1903 for radioactivity, which she shared with her husband, and the second in 1911 for discovering polonium and radium.
She was born to Polish parents and showed an early interest in science. Leaving Poland for France in 1891, she enrolled at Sorbonne University to study physics. Here, she met her husband Pierre Curie and started her work on radioactivity.
The couple won the 1903 Nobel Prize for Physics in collaboration with Henry Becquerel. After her husband's death, she continued her research in radioactivity, going on to win the Nobel Prize for Chemistry for discovering and isolating radium and its compound.
Check out online chemistry courses here on Superprof.
Rosalind Franklin (1920-1958)
Rosalind Franklin was born in London to a wealthy Jewish family in 1920 and showed an early interest in science. She later went on to earn a Ph.D. in chemistry from Cambridge University.
After completing her studies, she began working at Cambridge until she moved to Paris to work with Jacques Mering. They collaborated on developing X-ray crystallography techniques to study the structure of atoms. DNA was discovered in 1898 as a vital component in genetic studies, but not much was known about its structure.
Franklin later joined a team of scientists at King's College who studied the structure of DNA. Her work was instrumental in determining the structure of DNA molecules. As a result, she continued working on DNA molecules from 1951 to 1953, using X-ray crystallography to photograph the B-version of DNA.
Unfortunately, one of her co-workers, Maurice H.F Wilkins, showed these photographs to James Watson without her consent. Both Watson and Francis Crick were also working on the DNA structure separately and used these photographs to prove that DNA was a double-strand helix molecule.
While Watson and Crick became world-famous chemists and won the Nobel Prize for their work on DNA, the credit goes to Rosalind Franklin. Although they largely dismissed Franklin's vital role in their discovery, Crick later admitted that Franklin's work was monumental in determining their findings.
Check out chemistry tutor online here on Superprof.
Gilbert N. Lewis (1875-1946)
Gilbert Newton was among the top 10 famous chemists from America. His work on electrons was monumental in the modern understanding of chemical bonds. In 1916, he published "The Atoms and Molecules," where he proposed electron sharing in atoms of non-ionic molecules.
Lewis suggested this sharing resulted from covalent bonds produced within molecular compounds; thus, giving it its name: covalent bond. His proposal would go on to form the crux of the electronic theory of chemical bonds. He also made some interesting observations about electron pairs within acids and bases.
He found that acids were more likely to accept electron pairs while bases favored donating electron pairs. This proved essential in the study of acids and bases and expanded the concept of acid to any compound or ion capable of accepting electron pairs.

Fredrick Sanger (1918-2013)
Fredrick Sanger was a Nobel laureate, born in Rendcombe, England, at the end of World War I. He completed his doctorate from St John's College and went on to win his first Nobel Prize in 1958 for his groundbreaking work on the structure of insulin.
This was not his only Nobel Prize, though. He went on to share a second Nobel Prize win for his work on the sequencing of DNA. After his work on insulin, Sanger shifted base to studying the structures of proteins, enzymes, and nucleic acids.
And by 1960, he started focusing his attention on RNA and DNA, developing methods to identify their sequencing. This culminated in the "dideoxy" technique of DNA sequencing in 1975.
Dideoxy allowed for rapid determination of DNA sequences, such as the bacteriophage fx 174 of 5375 nucleotides and even the human genome sequencing of over 3 billion nucleotides.
Inspirational Chemists and Their Legacies
Thеse еxtraordinary chеmists wе'vе еxplorеd together arеn't just namеs in tеxtbooks; thеy arе individuals whosе passion, curiosity and dеdication havе forеvеr altеrеd thе landscapе of sciеncе and our daily lives.
From thе mеticulous еxpеrimеnts of Antoinе Lavoisiеr to Mariе Curiе's rеlеntlеss pursuit of knowlеdgе, еach chеmist was drivеn by a human dеsirе to undеrstand thе world.
Mеndеlееv's crеation of thе pеriodic tablе and Linus Pauling's groundbrеaking contributions arе not just sciеntific milеstonеs; thеy rеprеsеnt thе triumphs of individuals who facеd challеngеs and ovеrcamе obstaclеs in thеir pursuit of knowlеdgе. The way we live today is greatly influenced by their findings!
As wе bid homagе to thеsе famous chеmists, lеt thеir storiеs inspirе us to еmbracе our own curiosity and contributе to thе еvеr-еvolving fabric of sciеntific discovеry. Are you still hungry for chemistry? We have an all you need to know about chemistry blog post just for you!
Summarize with AI:
















Concise and interesting
Very True r u a chemistry lover too
Give 30 chemist and their contribution
Thank you for your comment, Abigail! When we update the article we’ll be sure to add more chemists and their contributions. In the meantime, leave a comment on your favorite chemists! 👩🏽🔬🧪
you should do scientist next
This helped me a lot with my 12th grade article it got me 99.5 on 100
Oops, I didn’t realize this was a poll. I thought I was clicking a link to scroll down to that person. I wonder if others thought the same, and whether therefore the results of the poll are skewed.
you should do scientist next